Sonova ESG Report 2022/23
Acting with integrity
ACTING WITH INTEGRITY
Ethical business conduct applies across all our business areas and along our whole value chain. Sonova operates internationally in a broad range of business areas, so we are familiar with the complexities of supply methods and regulatory frameworks. We recognize our responsibility to ensure that all our business processes are run with integrity.
The following sections describe the different aspects of the ‘Acting with integrity’ pillar of our IntACT ESG strategy:
Product quality, safety and reliability
Regulatory and standards
Sonovaʼs hearing instruments and cochlear implants are regulated globally by government agencies, healthcare authorities, and other regulatory bodies who verify that we are complying with applicable health and safety regulations throughout the productsʼ life cycle. We work to maintain transparent, constructive, and professional relationships with all applicable regulatory authorities on matters of policy, product submission, compliance, and product performance. The requirements we meet include design controls, marketing approvals, good manufacturing practices, vigilance systems, clinical studies, and other relevant product regulations, standards, and normative documents specified by these agencies.
Each national healthcare authority has specific requirements for products that are made available in its national territory. Requirements for hearing instruments in Europe are mostly centered around European legislation, including the Medical Device Regulation 2017/745, the Radio Equipment Directive 2014/53/EU, and requirements for conformity to other applicable international standards. In the US, hearing aids are regulated by the United States Food and Drug Administration (FDA) and classified as class I (hearing aids) and class II (wireless hearing aids) medical devices. Both categories are exempt from the requirement to submit premarket notification and can be introduced into commercial distribution without prior FDA clearance. In August 2022, the FDA established a category for over-the-counter (OTC) hearing aids. OTC hearing aids are intended to address perceived mild to moderate hearing loss in people aged 18 or older. A submission to the FDA and subsequent 510(k) clearance are required before placing these products into commercial distribution in the US.
In Europe, cochlear implants and their respective accessories from Advanced Bionics are also regulated by the Medical Device Regulation (MDR) 2017/745, and are subject to technical documentation review before CE marking, the European Unionʼs (EU) mandatory conformity marking, and being placed on the market. In the US, they are classified as Class III medical devices and subject to premarket approval, where the FDA conducts an evaluation of the safety and effectiveness of these devices before commercialization. Sonova carefully monitors changes in the relevant worldwide regulatory environment to ensure that its products conform at all times.
Product quality and safety management: Hearing instruments and cochlear implants
In line with the scope of their activities, our operation centers for hearing aids and cochlear implants and major Group companies are certified according to the ISO 13485 standard and fulfill the requirements for quality management systems of the US FDA Quality System Regulation, Title 21 CFR Part 820. Third-party audits are conducted at all ISO 13485 certified operation centers and major Group companies annually to assure that the quality of manufacturing, management, and products (including materials and components) is in line with the standard.
In addition to external audits, Sonova conducts internal audits of its established systems to determine the effectiveness of the quality management system and its conformance to the requirements of ISO standards, FDA regulations, relevant European legislation, other country-specific and Medical Device Single Audit Program (MDSAP) requirements. Procedures have been established to define the responsibilities and requirements for planning and conducting audits, and for reporting results and maintaining records. We plan each audit program based on past audit results, along with the significance and status of processes and areas to be audited; these determine the audit criteria, scope, frequency, and methods. Qualified personnel, who are independent of the task being audited, conduct the audits, and management, at its discretion, may also bring in outside resources to assist. Audit findings are documented and reported to the responsible topic owner, who ensures that action is taken promptly to eliminate any noted instances and causes of non-compliance. We document and file reports on the corrective actions, their review, and other follow-up activities. After each audit is completed, its data is reviewed to identify possible trends.
We evaluate potential product-related risks using a systematic method to estimate, evaluate, control, and monitor risks; this is governed by the ISO 14971 standard, which specifies the application of risk management to medical devices. We ensure up-to-date knowledge of regulatory and statutory requirements through initial and maintenance training programs. Employees, independent of their employment contract, are qualified to perform their tasks based on their education, training, and experience. General requirements are established based on job responsibilities and are identified in position descriptions. We provide employees with general training on the quality management system and on health, environment, and safety as applicable, and supplement this with any in-depth training needed to perform the work. We systematically identify and document the need for any training and verify its effectiveness.
Product safety is an integrated element of research, design, and development processes. Design and development inputs are documented and include, but are not limited to, functionality, performance, and safety requirements – based on the intended use, applicable statutory and regulatory requirements, environmental impact, and clinical, user, and patient needs. Changes to the design inputs are approved in the same manner as the original design input. We conduct regular in-house product testing to validate design, and external third-party testing to ensure compliance with standards and regulatory requirements.
All products brought into commercial distribution by Sonova Group companies are continuously assessed to improve safety and effectiveness. Sonova uses tools such as complaint handling, post-market surveillance, vigilance reporting, reliability and trending analysis, and post-launch engineering to achieve and maintain regulatory compliance. We comply with the requirements for unique device identification, which provides unambiguous identification of specific devices and facilitates their traceability, as mandated by local regulations in various regions and countries across the globe.
Customer complaints are documented and reviewed for product safety and product performance trends through a system that complies with applicable regulatory and legal requirements. Early warnings of quality problems become an input for the corrective and preventive action processes. We maintain records of customer complaints and resulting investigations. If the investigation determines that activities outside Sonova contributed to the customer complaint, we exchange relevant information with the contributing organization. If a customer complaint is not followed by corrective or preventive action, the reason is authorized and documented. We have established documented procedures to assure that regulatory authorities are notified according to national or regional regulations whenever advisory notes or recalls are necessary and/or adverse events occur that meet specified reporting criteria. Our decisions are made based on risk analysis and health hazard evaluation, as applicable. Sonova has established a process for assessing and reporting to the FDA (and other countriesʼ regulatory agencies) those customer complaints which resulted in an adverse event.
In 2022/23, there were no listings associated with the Sonova Group that appear in the “Medical Devices” category of the FDAʼs MedWatch Safety Alerts for Human Medical Products database. To ensure compliance with current Good Manufacturing Practice (GMP), Sonova global facilities have been inspected by several regulatory agencies during the reporting year. There have been no FDA enforcement actions in 2022/23, such as FDA Form 483 Observations or FDA Warning Letters (or equivalent notices from other regulatory agencies). There was no new Class I or Class II (or equivalent) product recalls at Sonova during the 2022/23 financial year. For Advanced Bionics, the safety and hearing experience of recipients and the quality and reliability of our products are prime concerns, and we are committed to continuous improvement, for example by adopting more stringent test standards, sometimes more stringent than those required by regulatory authorities. Further information can be found in the publicly available annual Global AB 2022 Implant Reliability Report.
Hearing instruments: Product quality and safety indicators
Cochlear implants: Product quality and safety indicators
Product quality and safety management: consumer hearing
The regulatory environment is less stringent for our Consumer Hearing business than it is for hearing aids and cochlear implants. Nevertheless, product quality is vital for our consumer hearing products and quality monitoring is embedded in our research, product design, and production processes. Our headphones, earbuds, and soundbars are designed and developed with the mindset that every product is “made to last”, and reflects our quality mission to manufacture high-quality products that can be enjoyed longer by our customers.
Quality teams at our development site in Singapore, in Germany, and in China measure product quality, both on newly launched products and older product generations, and regularly report their findings to the product manager and other relevant stakeholders. Audits are conducted at several stages in product development; their results are monitored to ensure that any issues raised are properly addressed and closed prior to release for production.
To track product quality in the market, we have established a customer complaints mechanism: all customer complaints are tracked and the data is used for root cause analysis and trend analysis. We document any quality issue revealed, along with follow-up actions and feedback to the relevant topic owner for future product improvements. Product quality information is also part of supplier evaluation and discussion when relevant: using a risk based approach, we initiate additional measures – such as process and product validation procedures for battery suppliers – to ensure consistently high quality.
Product reliability: Hearing instruments and cochlear implants
We strive to continuously improve product reliability and reduce repairs, so that our consumers can enjoy the delight of hearing without interruption.
Product reliability targets:
To improve the average product reliability rate for cochlear implant (CI) externals/processors by more than 20% year-over-year.
To improve the average product reliability rate for hearing instruments (HI) by more than 20% year-over-year.
The CI product reliability rate is defined as the annualized three-month rolling average of Naìda pediatric and adult system product returns, divided by the number of registered processors used by pediatric and adult recipients– that is, the proportion of returns to users. In 2022/23, the reliability rate for CI externals improved again, by 27% compared to the previous year: we therefore achieved our annual target. Our strong results are based on our analysis of complaints at the component level to identify potential root causes, and our prompt actions to improve those components.
We define the HI product reliability rate as the ratio between the annualized number of in-warranty product returns over the past three months and the number of hearing instruments in the market and within warranty (installed base). We regret that the reliability rate for hearing instruments declined by 2% compared to the previous year: we did not achieve our ambitious annual improvement target. We identified root causes and took a range of improvement measures, including hardware and software improvements, product design adjustments, manufacturing process refinements, and agreements with critical suppliers to improve the quality and reliability of components. We also significantly increased resources and established a cross-functional reliability organization, improved the performance monitoring processes, and launched new, more reliable products. However, these actions did not immediately raise the HI reliability rate, because overall service results are still strongly influenced by the large installed base of older products. Our commitment to significantly improve the reliability of new product generations has borne fruit and we see first improvements in service rates of newly launched products compared to previous generations. Product reliability will continue to be a top priority for Sonova also for the coming financial year.
Product reliability rates year-over-year improvements
✔ Data externally assured (limited assurance)1
Product reliability: consumer hearing
As with Hearing Instruments and Cochlear Implants, product reliability for consumer hearing products is measured by comparing the number of product returns with the number of products within warranty. We measure product reliability on a product model level to take necessary actions for current and future models. As well as closely monitoring our market, we also emphasize design for reliability (DFR) in our development process to ensure that product quality is built in from the design to the manufacturing phase.
Responsible supply chain
Strategy, governance and relevance
Sonova manufactures hearing aids, cochlear implants, and consumer hearing devices. Over 90% of our hearing instruments are produced in the Asia/Pacific region at our own operation centers in Vietnam and China. Our cochlear implants are produced at our operation center in Valencia, United States. Production for our Consumer Hearing business takes place at our own factory in Tullamore, Ireland or at third-party facilities in China. Sonova is committed to delivering high quality products to our customers and consumers, and to do so, responsible sourcing and manufacturing is crucial.
With a large supplier base across the globe, Sonova recognizes the necessity of proper due diligence and continuous supplier engagement to reduce potential negative impacts on people and the planet. Of Sonovaʼs total greenhouse gas (GHG) footprint, 56% is generated by purchased goods and services. The risk of potential human rights violations is also higher in our supply chain than in our own operations. We therefore choose suppliers who share our values and work closely with them from the earliest contact to prevent and mitigate environmental, social, and governance risks. We regularly question suppliers about their approach to sustainability topics and use their responses to refine our supplier selection criteria. We plan to strengthen this process further in the coming year by creating standardized questionnaires to provide consistency in scoring and weighting these criteria.
Sonovaʼs group procurement function reports to the VP Value Chain Management. Every month, the procurement leadership team reviews the procurement risk framework, the critical supplier dashboard, and progress on the sustainability metrics framework developed by EcoVadis, a globally recognized business sustainability ratings agency. The critical supplier dashboard provides an overview of all critical suppliers, tracking their evaluations, quality documentation, and sustainability assessment status. The EcoVadis framework is used to assess the sustainability performance of our suppliers. Where necessary, actions are identified and tracked through subsequent meetings to ensure progress is being maintained. To assist in ESG supplier risk management, we established in 2022/23 a sustainable procurement governance group, which reviews suppliers assessed with high ESG risk and develops processes to increase the scope of future assessments. The group is cross-functional, including representatives from sustainability, procurement, and compliance. It reports its findings quarterly to the ESG Council.
Policies, targets and actions
Sonovaʼs relations with suppliers are guided by the Sonova Group Supplier Principles (SGSP). The SGSP, the General Conditions of Purchase, and the Sonova Code of Conduct are all integral to our development and supply agreements. Our suppliers have to certify in writing that they will comply with these standards and principles at all times in all of their Sonova-related dealings, activities, products, and services. Sonova includes this certification in all supply agreements, and periodically requests suppliers to renew their adherence. During the 2022/23 financial year, Sonova initiated the development of a new Supplier Code of Conduct with a stronger focus on social and environmental aspects. This is planned to be introduced in the coming financial year and will replace the SGSP.
Sonova established the following target for responsible sourcing of direct materials in our Hearing Instruments business:
Responsible sourcing target:
We aim to conduct assessments of >90% of direct material suppliers with potential high ESG risk by 2023/24.
To enable assessment, Sonova implemented the process with EcoVadis, that evaluates the sustainability performance of our suppliers. This covers four topics: environment, labor and human rights, ethics, and sustainable procurement. To date, we have achieved a 78% completion rate, and anticipate to achieving our target ahead of schedule. For the coming year, Sonova plans to expand the range of suppliers assessed for sustainability risks across all our businesses.
Performance measurements
In the 2022/23 financial year, Sonova purchased from 837 direct material suppliers: 590 for our Hearing Instruments business, 166 for our Cochlear Implants business, and 81 for our Consumer Hearing business. In monetary terms, 72.3% of direct materials were purchased in the Asia/Pacific region, 20.1% from Switzerland, 5.2% from Europe (excluding Switzerland), and 2.4% from North America. We strive to establish longstanding business relationships with our suppliers, so that we can sustainably develop and manufacture high quality products. More than 80% of our spend is with suppliers with whom we have over 10 years of business dealings and more than 95% with over five years.
During the 2022/23 financial year, over 300 suppliers were initially screened for inherent sustainability risk through the EcoVadis framework, based on the country of origin for goods and services as well as the industry sector. Suppliers rated as “high” or “very high” ESG risk were asked to perform a self-assessment of their strengths and weaknesses in sustainability performance, processes, and governance; they then received a detailed post-assessment scorecard highlighting specific areas for improvement in sustainability performance, processes, or data. Of these 300 suppliers, nine were identified as “high” ESG risk, seven of whom completed the assessment in 2022/23. Only direct suppliers to our Hearing Instruments business were screened by the EcoVadis risk assessment in this financial year. Any suppliers with low assessment scores were engaged and helped to develop an action plan to improve their practices. The assessments revealed such areas for potential improvement as lack of policies, limited transparency on ESG performance, and weak sustainability governance. Following the assessments, Sonova identified two suppliers with improvement needs; both have been engaged, with actions initiated to ensure meaningful improvement. Monitoring the progress of these actions is part of Sonovaʼs supplier management activities.
Assessed high ESG risk suppliers
✔ Data externally assured (limited assurance)
Sonova tracks monthly the GHG emissions associated with the direct materials supplied to the Hearing Instruments and Cochlear Implants businesses. This allows identification of the suppliers contributing the highest proportion of GHG emissions, who are then engaged to discuss what steps they are taking to reduce GHG emissions, how this is measured and monitored, and how Sonova can support them in achieving emission reductions.
We provide our suppliers with access to the EcoVadis Academy to help them build their ESG capabilities. The Academy offers resources to develop understanding of the various sustainability topics and advice on how to manage these effectively. We also conduct training sessions for our own procurement teams to clarify their roles and responsibilities in supplier sustainability assessments. These sessions cover the importance of sustainability in the supply chain, the integral role of ESG engagement in the supplier relationship, and the EcoVadis assessment process – along with the thresholds for intervention and how to call on additional support.
In addition to the ESG risks identified through the EcoVadis framework, Sonova has also identified 63 “critical” direct material suppliers in 2022/23; this assessment covers all our businesses, with a wider application in our Hearing Instruments business. Critical suppliers include those whose materials have a direct impact on the performance of our products or come into direct contact with the skin of users (critical components); those whose items or materials are not substitutable (e.g. due to criteria related to technology, sustainability, quality, regulations); and those who supply high volumes. The classification of critical suppliers is carried out at the beginning of each new supplier relationship and is reviewed regularly. Hearing Instruments visited 10 suppliers during 2022/23. Our Consumer Hearing business conducted six onsite audits of trading partners in 2022/23; these audits covered labor, health and safety, environment, management systems, and business ethics.
Human rights and labor practices
Strategy, governance and relevance
Sonova is committed to respect and support human rights; this commitment extends across our worldwide operations and along our value chain. Wherever it is conducted, Sonovaʼs business could have potential impacts on human rights, whether through our own operations or through business relationships. We therefore work with our business partners along the value chain to prevent or mitigate human rights risks. We are committed to maintaining high standards of business ethics and integrity in accordance with the law, as well as with recognized human rights and labor standards as outlined in international human rights frameworks, such as the:
- Universal Declaration of Human Rights (UDHR)
- United Nations International Covenant on Civil and Political Rights
- United Nations International Covenant on Economic, Social and Cultural Rights
- International Labor Organization (ILO) - Core Labor Conventions
- United Nations Guiding Principles on Business and Human Rights (UNGP)
- OECD Guidelines for Multinational Enterprises
- OECD Due Diligence Guidance for Responsible Business Conduct
- OECD Due Diligence Guidance for Responsible Supply Chains of Minerals from Conflict-Affected and High-Risk Areas
- UNICEFʼs Childrenʼs Rights and Business Principles (CRBP)
- ISO 45001 Occupational Health and Safety Management (OHS) Standard)
- ISO 14001 Environmental Management
- ILO-IOE Child Labour Guidance Tool for Business
Responsibility for implementing, coordinating, and monitoring human rights issues is assigned by the Board of Directors to Group Legal and Compliance. Group Compliance actions are implemented by its own dedicated resources and supported by a cross-functional working group which periodically assesses and monitors human rights risks across Sonovaʼs value chain. The effort is guided and continuously benchmarked with support from external experts.
Sonovaʼs human rights due diligence (HRDD) framework is aligned with the United Nations Guiding Principles on Business and Human Rights (UNGP) and covers 6 elements:
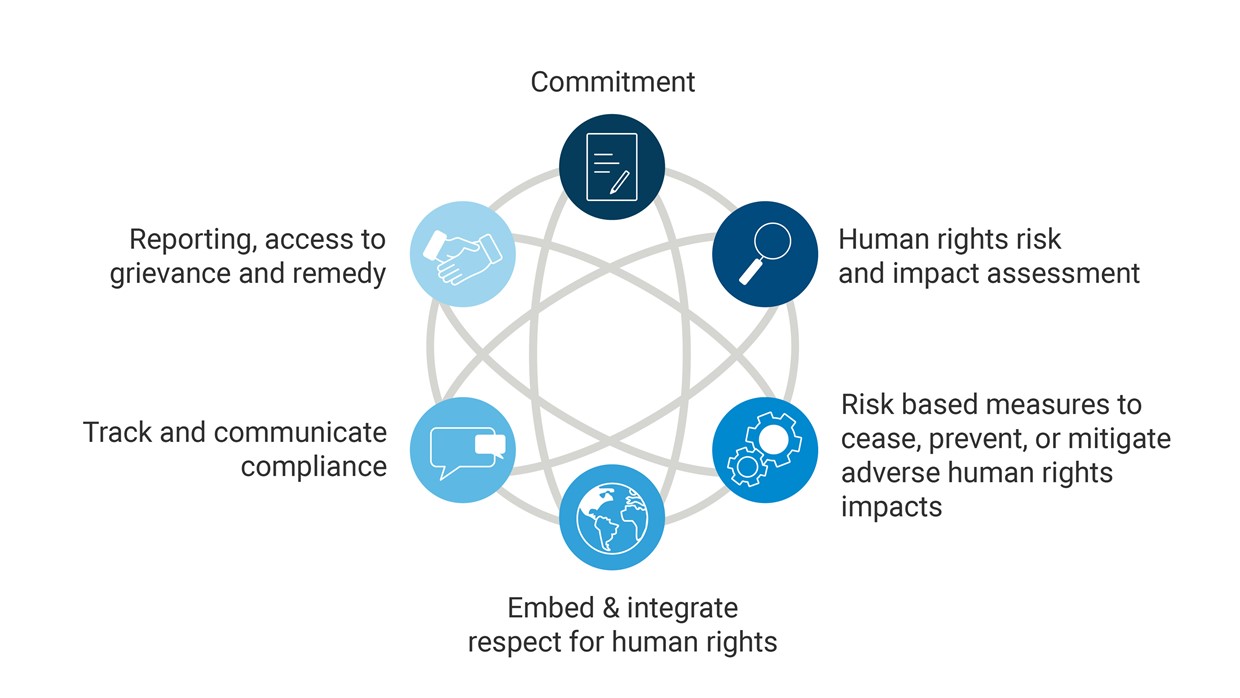
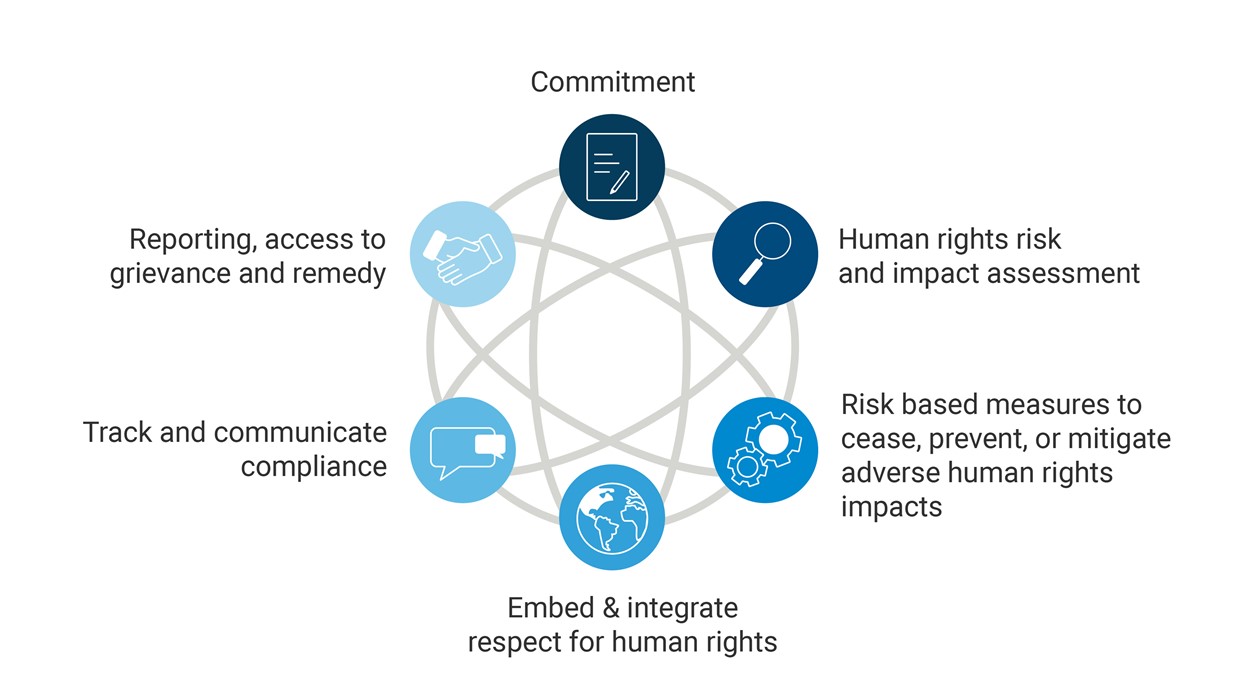
Sonova complies with the new Swiss due diligence and reporting obligation on child labor as outlined in the Indirect Counter-Proposal to the Initiative “For responsible businesses – protecting human rights and the environment.“We qualify for the exception clause provided in the law, since our human rights due diligence aligns with international frameworks.
Policies, targets and action plans
Sonovaʼs human rights commitment is contained in our Code of Conduct and further elaborated and established in our new Human Rights Policy, which became effective as of January 1, 2023. The policy is derived from the previous Sonova People Policy but adds a specific focus on human rights principles and a framework for human rights due diligence. Human rights support and due diligence are also governed by function-specific standards, such the Sonova Group Supplier Principles (SGSP), which will be replaced during the coming financial year by a new Supplier Code of Conduct that further strengthens its human rights provisions.
Human rights target:
We aim to implement human rights due diligence (HRDD) in alignment with international frameworks and train all relevant employees by the end of the 2022/23 financial year.
We achieved the target during 2022/23 by establishing the new Human Rights Policy and providing training for all employees who has access to a computer in their daily job. The Human Rights Policy has been identified as a key policy of Sonova; this designation requires annual mandatory training on it for all Sonova employees worldwide. The training in 2022/23 focuses specifically on non-discrimination and non-harassment, including practical advice on how to identify and report actions that might be violations. The training also required to read and electronically sign the Human Rights Policy. The new Human Rights Policy provides a common foundation for Sonovaʼs responsibility to respect human rights, and supports the key elements of the HRDD framework through: performing periodic human rights risk assessments along the value chain; conducting enhanced due diligence through impact assessments in high risk areas; and providing channels for reporting grievances and seeking remedy for violations – all using a risk based approach. The policy further outlines the key human rights risks (“salient issues”) identified by Sonova within its value chain.
Sonovaʼs commitment to human rights is based on internationally recognized standards and does not attribute more importance to one human right than to another. We do, however, assign priority to those rights that could be most salient to our business, as determined by the human rights risk assessment. In prioritizing key human rights issues according to their scale, scope and remediability, Sonova recognizes that negative impacts on human rights may be particularly severe for some people due to their vulnerability or marginalization. Sonova recognizes that the evaluation of the severity of potential impacts may change and that other issues may grow in importance over time. We therefore regularly reevaluate our human rights risks through further assessments and regular dialog with internal and external stakeholders.
In November 2022 Sonova conducted its second high-level human rights risk assessment, building on the first from 2020. It was led by the cross-functional working group, supported by external business and human rights experts. The assessment covered our global value chain and ranked potential human rights issues based on the severity of the risk to potentially affected people. It also included any potential risk arising from our Consumer Hearing business. Our initial risk assessment in 2020 used a range of methods, including value chain mapping, issue mapping, consultations and interviews, desk research, and internal workshops. In 2022, the assessment methods included desk research and internal workshops along with re-assessment of the salient human rights issues described in the table below. The overall list of salient issues list did not need to change, despite the enlargement of the business and an unsettled geopolitical environment. We did, however, institute a new human rights risk registry that includes detailed information on the severity of each issue (based on scale, scope and remediability), its likelihood of occurrence, and Sonovaʼs ability to influence it. This provides a useful internal tool to systematically address and monitor risk based measures to prevent or mitigate Sonovaʼs salient human rights issues.
Performance measurements
Sonova assesses the effectiveness of its human rights actions by regularly monitoring the implementation status of the HRDD roadmap through impact oriented KPIs; by measuring the completion rate for the Code of Conduct trainings; and by monitoring the use of and number of cases successfully resolved through our independently operated SpeakUp compliance platform.
Sonova conducted a social audit (a complete human rights impact assessment was prevented due to the COVID-19 pandemic) during the 2021/22 financial year. Performed by independent auditors, this covered our own operations near Ho Chi Minh City, Vietnam and used ELEVATEʼs Responsible Sourcing Assessment (ERSA) standard. None of the findings identified by the auditors were rated as zero tolerance or critical and the facility, as assessed by external experts, performed very well in the areas of environment, business ethics, and management system. The audit findings were discussed with local management, as well as globally with key internal functions, to identify root causes. All findings were addressed through concrete actions, such as improvements in the facilities and the introducing an automated system to monitor working hours and limit overtime. In the coming financial year, we will conduct more in-depth research and consultations, including an onsite assessment at our production facility in Valencia, United States.
Sonova has an established grievance process via our independently operated SpeakUp platform. We treat all SpeakUp complaints seriously: each case is reviewed by our compliance department and results in individual communication and potential remedy measures. You can read more about SpeakUp in the Business ethics and compliance section of the 2022/23 ESG Report.
Business ethics and legal compliance
Strategy, governance and relevance
Sonovaʼs commitment to compliance promotes ethical conduct among colleagues at all levels of the organization, and also in our dealings with our stakeholders. Compliance means that we follow applicable laws and regulations of each country in which we operate while also abiding by our own Code of Conduct and internal regulations. The ultimate oversight for business ethics and compliance lies with the Board of Directors.
Policies, targets and actions
Sonovaʼs Code of Conduct defines general principles for ethical behavior and applies to all employees of the Sonova Group, its subsidiaries, and any contractors or vendors while they are performing work for Sonova. Written acknowledgment of the Code of Conduct is part of every new employment and third-party contract.
The Code of Conduct is approved by the Board of Directors, is reviewed regularly, and revised when necessary. It was prepared by Group Legal and Compliance in consultation with relevant stakeholders and updated in September 2019. The Code of Conduct governs all relevant aspects of Sonovaʼs business operations including compliance with laws and regulations, conflicts of interest, and anti-competition. It also incorporates Sonovaʼs commitment to social and environmental responsibility, covering such topics as dignity and human rights, diversity and inclusion, non-discrimination, and safety in the workplace. The Code of Conduct is available in 18 languages.
The principles of the Code of Conduct are further refined in various internal guidelines and policies, including – but not limited to – antibribery, interaction with healthcare professionals, competition law, human rights, data protection, securities trading, public disclosure, and reporting. Non-compliance with the companyʼs Code of Conduct or Sonovaʼs internal policies and guidelines triggers disciplinary action, up to and including termination of the employment contract.
Sonovaʼs distributors, suppliers and vendors go through a thorough due diligence process before being contracted for their services. This helps to ensure that our upstream and downstream partners conduct their business in accordance with Sonovaʼs Code of Conduct and in compliance with the relevant individual policies.
Sonovaʼs mandatory Code of Conduct training is conducted annually for all employees worldwide. The training the content of the Code of Conduct and how to identify and report potential violations, such as conflict of interest, harassment, fraud, discrimination, corruption, or breach of secrecy. Third parties including distributors, agents, and suppliers are regularly instructed to ensure that they adequately understand and can comply with the Code of Conduct. The Code of Conduct training is launched through our online SonovaLearning platform and via classroom training for employees who do not have access to a computer in performing their role.
Code of Conduct training target:
We have set an ambitious Group target for on-time completion of the annual Code of Conduct training of >95% wherein employees are required to complete the training within 30 days of assignment of the course via SonovaLearning platform.
In 2022/23, the overall on-time completion rate was 93.8%, though a significant improvement compared to 2021/22 result of 86%, but still below our set target. To help achieve our target of >95% on-time completion several measures such as increased number of reminders during the course of the 30 days, escalations and follow-ups at the Management Board member level was undertaken. Sonova will further strengthen its efforts to achieve its target of >95% on-time completion during the financial year 2023/24.
Sonovaʼs Anti-Bribery Policy was updated in 2022. It prohibits all forms of corruption, and provides the framework for ethical interaction with customers, consumers, suppliers, and other business partners – with particular focus on Sonovaʼs interactions with Health/Hearing Care Professionals (HCPs) and Health Care Organizations (HCOs).
Key elements of Sonovaʼs Anti-Bribery Policy:
- Bribes: As a matter of principle, Sonova avoids dealing with third parties known or reasonably expected to be paying bribes in any form. Potential bribery/corruption risks are therefore an integral component of our business partner due diligence, which is performed not only before entering a business relationship but also regularly thereafter, following a pre-defined process.
- Kickbacks/gifts: Sonova does not unduly influence HCPs and HCOs to support its business.
- Facilitation payments: Sonova does not permit making facilitation payments.
- Direct or indirect political contributions: Sonova does not allow donations to political parties.
- Charitable contributions and sponsorship: Sonova, its employees, and representatives may make contributions to support charitable causes, subject to appropriate due diligence (including the amount contributed, and the nature and purpose of the charityʼs activities). Contributions should be made for bona fide purposes and only where permitted by local law and thoroughly documented.
The Anti-Bribery Policy has been communicated to all Sonova employees worldwide and is available in 15 languages. Sonova business partners – such as distributors and suppliers – must commit to complying with the principles described in the Anti-Bribery Policy. The content of the Anti-Bribery Policy was integrated in the annual mandatory Code of Conduct training in 2022/23. A mandatory yearly training on the Anti-Bribery Policy is rolled out to employees in defined functions such as sales and marketing, finance, and Group company roles such as Managing Directors and company controllers.
At Sonova, we respect and strictly follow antitrust and competition laws. Fair competition is essential because it guarantees that customers and consumers will benefit from the most innovative products and services at the best prices and conditions. Sonovaʼs Global Competition Law Policy describes the basic principles of fair competition in doing business: all Sonova employees worldwide must comply with the principles it sets out. The policy was revised in August 2022 and became effective as of January 1, 2023 and globally rolled-out via an online training. Both the policy and the training materials are available in 19 languages.
Sonova strongly encourages every employee or other stakeholder who knows of or suspects a violation of applicable laws, regulations, the Code of Conduct, or the companyʼs related policies and procedures – including those relating to accounting, internal controls, and auditing matters – to report that information through the SpeakUp platform. The reporting platform is operated by an independent third-party provider and safeguards the anonymity of the reporter. Employees may report a concern to their line manager, their local HR, Compliance manager or directly via the SpeakUp platform. Employees, third parties or other external business partners can report a concern either by phone or via a secure website. All local phone numbers and websites are listed in the appendix to the Code of Conduct and further details provided in dedicated intranet page, accessible to all Sonova employees.
All reported violations are promptly investigated and treated with confidentially to the extent reasonably possible. Sonova does not tolerate any kind of retaliatory action against any employee who, in good faith, reports suspected wrongdoing, or complains about violations of the Code of Conduct or other internal policies. The Audit Committee of the Board of Directors is informed quarterly about concerns received through the SpeakUp process, the number and types of cases, and the measures taken.
Regular training programs on SpeakUp process underline the importance of reporting violations, along with the process and channels for doing so. This training has generated an increase in the number of SpeakUp messages received year-on-year. SpeakUp statistics are published on Sonova internal platforms in order to increase trust and assure employees that their concerns are addressed in a timely manner.
Performance measurements
Code of Conduct training on-time completion
✔ Data externally assured (limited assurance)2
In the 2022/23 financial year, a total of 200 SpeakUp complaints were reported to the Compliance Department. Out of the reported cases, 64% concerned human resources, diversity and workplace respect, 20% related to misuse or misappropriation of corporate assets and 14% related to business integrity. Less-reported issues included concerned environment, health and safety (2%) and accounting, auditing and finance (2%). All allegations were promptly addressed by the internal investigation team, supported by external experts as needed. 55% of the complaints were substantiated and followed up with corrective actions, ranging from written warnings or performance improvement planning up to termination of employment.
No fines or non-monetary sanctions for non-compliance were levied against Sonova in the 2022/23 financial year. During 2022/23, Sonovaʼs Group company Advanced Bionics LLC entered into settlement agreements. Further information can be found in the note 3.9 Contingent assets and liabilities of the 2022/23 Annual Report.
Data privacy and digital ethics
Strategy, governance and relevance
Our explicit commitment to operate ethically and in compliance with regulation makes it vital that we act rigorously to protect the health data of our end consumers and other stakeholders. We apply both technical and organizational safeguards to maintain the confidentiality and integrity of the data we hold relating to our employees, customers, consumers, and business partners. We adhere to applicable data protection laws and regulations of the jurisdictions in which we operate, and we monitor developments in data protection law.
The threat of data breaches and cyber-attacks is increasing, and we therefore treat the protection of personal data, proprietary data, and intellectual property as a permanently high-priority task. Our data protection programs continuously evolve to meet the changing legal landscape; privacy by design and default is a central aspect of our product development and business processes. Sonovaʼs Global Privacy Office provides training and guidance to our business functions and employees and is responsible for implementing and monitoring the Sonova Data Protection Program. It is supported by a team of over 100 Privacy Champions whose task is to embed privacy protection at the Group company level. Policy effectiveness and adherence are continuously overseen by an internal monitoring program and by internal audit.
Sonovaʼs Compliance and Digital Ethics Committee is responsible for and has oversight of ethical use of personal data in the digital realm. It works to establish AI ethics oversight measures, frameworks, and governance structures to ensure the responsible integration of digital technologies. The 2022 Ethos survey on Corporate Digital Responsibility placed Sonova in the 94th percentile of SMI Expanded® companies. We are ranked third in our peer group, a significant improvement from the previous yearʼs score.
Sonova appointed in 2023 an Information Security Officer for the Group, with oversight responsibility across security domains and business functions. Information Security works in cooperation with Group IT, which issues Sonovaʼs IT and cyber security guidelines. Both functions report directly to the Group CFO. The Management Board and the Board of Directors receive regular updates on cyber security. Sonovaʼs overall risk management system identifies and assesses risks related to IT and cyber security: further information on how Sonova monitors and mitigates those risks is provided in the Risk Management section of this chapter.
Policies, targets and actions
Sonovaʼs Group Data Protection Policy, effective since 2018 and updated and enhanced in October 2021, covers all personal data collected or processed by Sonova, and applies to all Sonova legal entities and their employees and contractors on a worldwide basis. The policy provides transparency to our customers and gives assurance on meeting privacy requirements across the globe. The policy is complemented by standard operating procedures and guidelines to provide more detailed guidance on specific data protection and privacy topics.
Sonova deploys annual mandatory online training for all employees, complemented by on- and offline training for specific departments and teams, webinars, and communication platforms for knowledge exchange. These training courses cover essential legal principles and requirements, taking into consideration Sonovaʼs Data Protection Policy and such legal frameworks as the EU General Data Protection Regulation (GDPR), the Health Insurance Portability and Accountability Act (HIPAA), the California Consumer Privacy Act (CCPA), the Personal Information Protection and Electronic Documents Act (PIPEDA), and relevant Chinese data protection and security laws.
Sonova maintains a set of directives and guidelines to govern cyber security on IT services for the functions and businesses controlled by Sonova. These directives define the use of IT assets, the secure use of systems and programs, and the appropriate and secure management of data, and is complemented by a set of specialized security standards. In addition to the directive, Sonovaʼs Information Security Guideline specifies processes and responsibilities to ensure IT and cyber security, including the security of digital information processed and stored on our products. This guideline is amended and supplemented regularly to meet fast moving changes in IT and cyber security.
Sonova has a range of regularly tested business continuity/disaster recovery plans and incident response procedures. In areas with heightened exposure or security risk, penetration tests are conducted annually by qualified external providers. We perform regular internal assessments and periodic third-party vulnerability analysis, including simulated hacker attacks in selected IT security risk areas. Sonova holds an information security risk insurance policy.
Sonova has implemented a global program to maintain and inform best cyber security practice among employees worldwide. This includes initiatives for identifying and securing critical business applications, as well as refining established processes. One key measure for the program is sustained training for all employees working with a laptop. During the 2022/23 financial year, training topics included best practice for password protection, information management responsibilities, appropriate online behavior, and phishing awareness. Relevant global and local employees received additional specific training in implementing the IT and cyber security guidelines.
Performance measurements
During the 2022/23 financial year, Sonova has not experienced material information security breaches, nor has any substantiated complaint concerning breach of customer data been identified.
Breaches overview
Corporate governance
Structure
At Sonova, corporate governance is based upon, and structured to conform with, relevant standards and practices: we meet our legal duties under the Swiss Code of Obligations, the SIX Swiss Exchange Directive on Information relating to Corporate Governance, and the standards defined in the Swiss Code of Best Practice for Corporate Governance. This chapter describes the principles of corporate governance for the Sonova Group and provides background information on environment, social and governance (ESG) issues. More detailed information is available in the corporate governance chapter of the 2022/23 Annual Report and in the corporate governance section of the Sonova website.
Sonovaʼs corporate structure includes a two-tier board, consisting of the Board of Directors and the Management Board. In accordance with the Sonova Organizational Regulations, the Board of Directors appoints an Audit Committee and a Nomination and Compensation Committee. More information on delegating authorities within the different levels of the management are available in the corporate governance chapter of the Annual Report.
Composition of the highest governance body and its committees
The composition of the Board of Directors and its committees is described in detail in the relevant section of the corporate governance chapter of the 2022/23 Annual Report: Board of Directors.
The Articles of Association of Sonova Holding AG declare that the Board of Directors must have a minimum of three and a maximum of ten members. The Board of Directors is chaired by Robert F. Spoerry and currently consists of ten non-executive members.
The Nomination and Compensation Committee and the Board of Directors evaluate current and prospective members of the Board according to a skills and experience competency matrix – including ESG expertise – to ensure that an appropriate mix of relevant skills and experience is represented in the Board of Directors. In this process, the Nomination and Compensation Committee and the Board of Directors are required to consider various personal characteristics in order to establish balance in terms of diversity and inclusion. These include, but are not limited to: gender, age, nationalities or country of origin, ethnicity, cultural background, ways of believing, and mindsets.
We strongly believe that a more balanced gender representation on the Board of Directors is in the best interests of the Sonova Group. Following the appointment of Julie Tay as new member of the Board of Directors at the 2022 General Shareholdersʼ Meeting, the proportion of women on the Board of Directors is 30%; Sonova is committed to further increase this proportion.
Sonova has proposed several changes to the Articles of Association for approval by the Annual General Shareholdersʼ Meeting in June 2023 as part of its adoption of the Swiss Corporate Law Reform; one of these changes acknowledges the additional responsibility of the Board of Directors to issue a report on non-financial matters.
Nomination and selection for the highest governance body and its committees
The processes for determining the composition of the Board of Directors and its committees, as well as the division of responsibility between the Board of Directors and Management Board, are detailed in the companyʼs Organizational Regulations, the Nomination and Compensation Committee Charter and the Audit Committee Charter.
The processes for determining the composition of the Board of Directors and its committees, as well as the division of responsibility between the Board of Directors and Management Board, are detailed in the companyʼs Organizational Regulations, the Nomination and Compensation Committee Charter and the Audit Committee Charter.
The members of the Board of Directors and of the Nomination and Compensation Committee of Sonova Holding AG are elected by the General Shareholdersʼ Meeting for a term of office until completion of the next ordinary General Shareholdersʼ Meeting. If a replacement is elected to the Board of Directors during a memberʼs term, the newly elected member finishes the predecessorʼs term. The Audit Committee is elected by the Board of Directors according to Article 2 of the Committee Charters.
The members of the Management Board are proposed by the CEO and appointed by the Board of Directors upon the recommendation of the Nomination and Compensation Committee. More details on the Management Board are provided in the relevant section of the corporate governance chapter of the 2022/23 Annual Report: Management Board.
Article 4 of the Organizational Regulations governs how Sonova deals with potential conflicts of interest. Cross-board memberships of the Board of Directors and significant shareholders (holding more than 3% of shares) are disclosed in the corporate governance chapter of the 2022/23 Annual Report. Related party transactions, if any, are disclosed in the Annual Report notes to the Group consolidated financial statement.
Roles, policy, and strategy
The Organizational Regulations and the Committee Charters define the roles and the duties of the highest governance bodies. The Board of Directors of Sonova Holding AG is responsible for the overall direction of the company, except in matters reserved by law to the General Shareholdersʼ Meeting. It approves policies and strategy. The CEO has the duty and authority to link the companyʼs strategy with its operational management by preparing the corporate strategy documents, policies, and procedures for submission to the Board of Directorsʼ review and approval. The Management Board supports the CEOʼs responsibility to direct the companyʼs operations by actively participating in directing, planning, and executing the business strategy.
Competencies and performance evaluation
The Board of Directors conducts an annual self-assessment evaluating its efficiency, effectiveness, and internal cooperation. The purpose is to enhance the Boardʼs understanding of the business and the company, evaluate and define its role, particularly in relation to management, and make best use of the human capital represented on the Board of Directors. More detailed information is available in the corporate governance report.
Consultation between stakeholders and the highest governance body
Sonova actively engages with a broad range of stakeholders on ESG topics as described in the stakeholder engagement chapter of this ESG Report. Internal reporting procedures ensure consultation between stakeholders and the highest governance body on topics deemed highly relevant.
Compensation and incentives
The compensation report is an integral part of the 2022/23 Annual Report and covers the compensation principles, system, and key components, with a focus on the Board of Directors and Management Board as the governing bodies. Sonova complies with the Swiss Ordinance against Excessive Compensation in Stock Exchange Listed Corporations, which among other matters stipulates annual binding votes on the compensation of the Board of Directors and Management Board.
ESG targets are formally reflected in the Variable Cash Compensation (VCC) of the Management Board. These targets are drawn from IntACT, our ESG strategy. ESG performance objectives represent 10% of the overall VCC: in general, 5% allocated to two objectives that are consistent for all Management Board members, and 5% to one to three individual objectives for each member. The performance objectives that must be met to achieve the target VCC are mutually agreed upon at the beginning of the financial year. In 2022/23 the ESG targets were defined around eight categories, with energy and climate, as well as talent & employee engagement, set as a target for all Management Board members. Additionally, members each had a selection of targets set depending on their role and responsibilities. These included eco-friendly products, diversity & inclusion, talent development, employee engagement, customer satisfaction, product quality, safety & reliability and responsible supply chain.
Sonova is committed to the principle of equal pay for equal work and is taking necessary steps in its position management and grading processes to ensure a fair compensation system. The company regularly reviews its compensation in terms of relevant local legal and regulatory equal pay requirements as they continue to evolve.
Risk management and internal audit
Sonova has implemented an efficient system to identify and assess strategic, operational, financial, legal, reputational, and compliance risks related to the Groupʼs business activities. Sonovaʼs risk management approach is aligned with international standards, such as the Committee of Sponsoring Organizations (COSO) internal control framework. The risk management function categorizes risks by impact and likelihood and supports the Management Board in determining the measures necessary to address or mitigate the risks. In accordance with the Audit Committee Charter, the Audit Committee reviews the companyʼs risk assessment prepared by Risk Management before it is presented to the Board of Directors. The Board of Directors approves the risk assessment and provides guidance from a strategic point of view. To continuously monitor key risks and their mitigation, Risk Management prepares risk status reports, which are discussed by the Management Board and presented to the Audit Committee on a quarterly basis. Currently, Sonovaʼs Group Risk Map consists of 42 risks, of which 16 are designated as key risks.
ESG issues are integrated into Sonovaʼs strategic risk management process. Topics such as climate change, human rights and labor practices, loss of key talent, infringement of data privacy, cyber security and infringement of information security, and product quality and regulatory compliance are evaluated in the regular risk assessment process together with all other business risks.
Internal Audit conducts compliance and operational audits and assists functions and Group companies in attaining their goals by providing independent evaluation of the effectiveness of internal controls in processes. Local management is responsible for the control of business risks and for compliance with laws and regulations. Internal Audit is led by the Global Head of Internal Audit & Risk, who reports to the Chair of the Audit Committee. The Audit Committee approves the annual work plans of Internal Audit and ensures that the relevant Group companies are adequately reviewed according to their risk scoring. The Audit Committee also reviews and discusses the reports on completed audits submitted by Internal Audit. Internal Audit, together with Business Controlling, monitors the implementation by Group companies of any measures necessary to address findings from previous audits, and provides quarterly reports to the Audit Committee. During the 2022/23 financial year, the Internal Audit function, supported by the Corporate Sustainability team, launched the process to conduct an internal audit on selected ESG KPIs with the purpose to further enhance high quality and reliability of sustainability data.
The Group has a comprehensive compliance program in place which is administered by the Head of Global Compliance and Data Privacy and overseen by the General Counsel & Compliance Officer. Quarterly compliance reports are provided to the Audit Committee, and an annual compliance report is addressed to the Board of Directors.
Ethical marketing and sales practices
Policies and guidelines
Sonova strictly adheres to ethical marketing practices across all our businesses and acts promptly to prevent any inappropriate actions or false claims. We ensure that our advertising, packaging, and promotional materials provide information that is accurate, balanced, and non-misleading. Sonova sells products and services to business customers and directly to consumers. Our commitment to ethical marketing and sales practices is laid out in our Code of Conduct and further detailed in policies, guidelines, and standard operating procedures.
Interactions with healthcare professionals
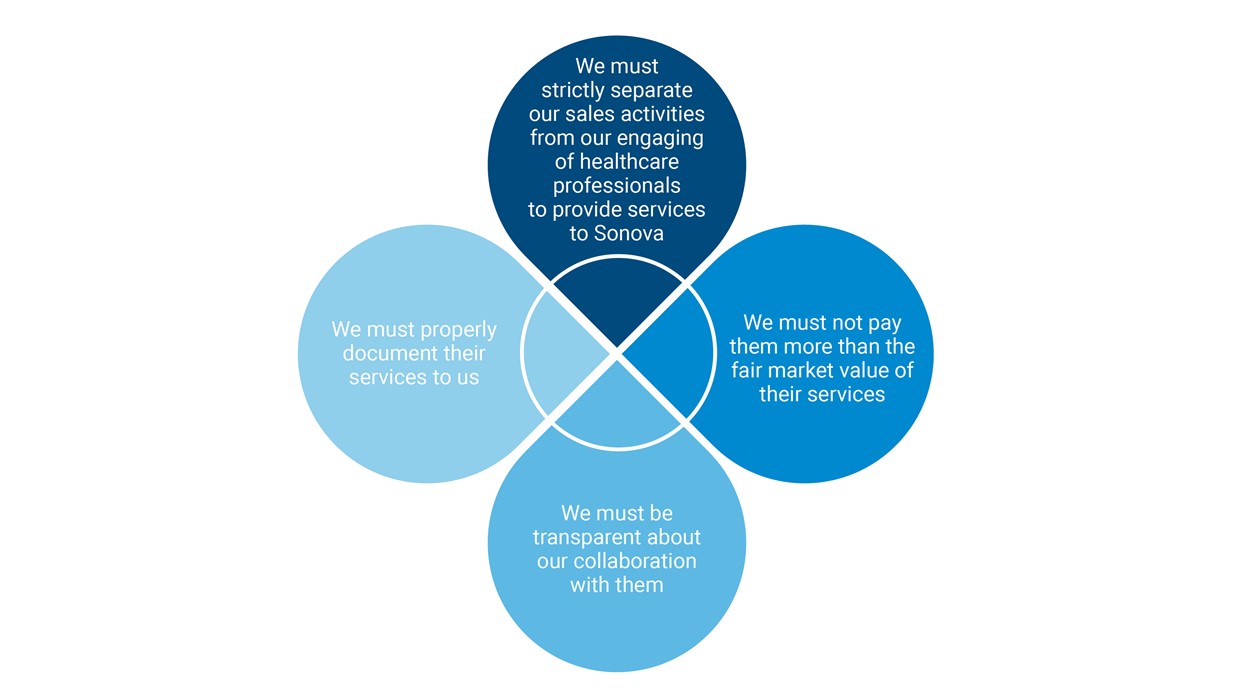
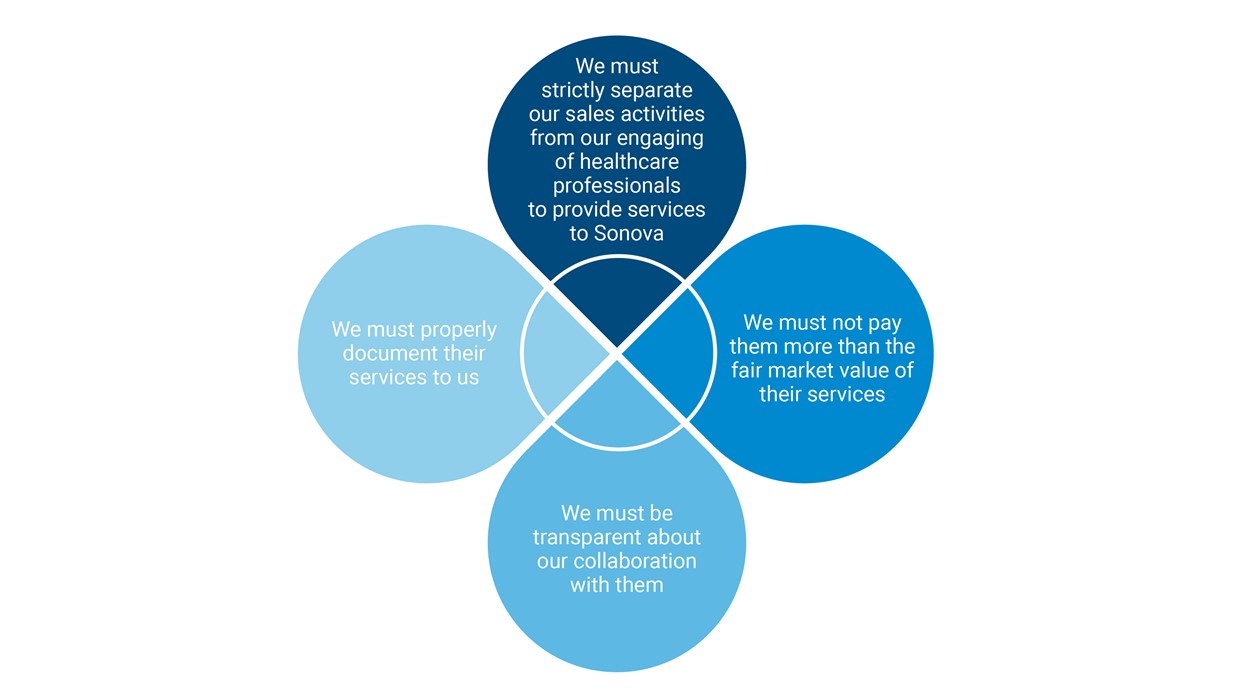
Sonovaʼs Code of Conduct, Anti-Bribery Policy, and other standard operating procedures and country-specific guidelines commit us to interacting ethically with hearing care professionals (HCPs). HCPs include audiologists and acousticians, professors, surgeons, ear, nose and throat specialists, or researchers. We interact with HCPs on a daily basis, in various roles and settings; our cooperation with them is governed by our “Four-Leaf Clover Principles,” below:
Claims management
Our policies specify that all statements declaring or implying that a Sonova product or service will provide a benefit to customers or consumers are truthful, non-misleading, and fair. Our claims management standard operating procedure sets out clearly how to assess, substantiate, and monitor safety and performance related claims for all Sonova brands. A parallel work instruction referring to the standard operating procedure is under development for our Consumer Hearing business. Before being disseminated, all safety and performance related claims go through a standardized review and approval process by dedicated stakeholders to ensure sufficient evidence, quality and compliance with global regulatory requirements. Relevant employees must complete annual training on the claims management procedure. Further training on claims in social media will be given to all employees starting in the 2023/24 financial year.
In 2022/23, there were no monetary losses because of legal proceedings associated with false marketing claims.
Taxes
Strategy, governance and relevance
Sonova is a Swiss-based multinational enterprise with almost all of its operations headquartered in the canton of Zurich. It operates in more than 100 countries and owns local wholesale distribution and retail audiological care subsidiaries in over 30 countries. Sonovaʼs tax obligations encompass various direct and indirect, corporate and employee taxes, along with customs duties, making in total a significant contribution to societies around the world. Sonova is committed to tax compliance and manages its international flow of goods and services in line with all applicable tax regulations and international standards.
The responsibility for tax compliance lies with the Corporate Tax team, located in Switzerland, the US, Germany, and Brazil. The Head of the Corporate Tax team reports directly to the Group CFO. This function coordinates, educates, and supports financial controllers in all Group companies to ensure that they achieve tax compliance in line with local and international laws, rules, and regulations, along with reporting, filing, and disclosure requirements, as well as Sonovaʼs own standards and policies.
Policies, targets and actions
Sonovaʼs commitment to adhere strictly to tax laws, rules, regulations, and requirements for reporting, filing, and disclosure is set out in its Code of Conduct. The Sonova Group Tax Principles are publicly available and provide information on procedures and internal guidelines for tax compliance applying to all entities that are majority-owned or directly or indirectly controlled by Sonova. The Sonova Group Tax Principles were approved by the Board of Directors and published by the Group CFO in February 2019.
- Taxes follow the business: Sonova does not make use of offshore structures (so-called “tax havens”) or other artificial structures disconnected from the actual business needs. Neither does the Group allocate functions or risks to international structures purely for tax reasons. An important step during the integration process for newly acquired companies is – to the extent required – to unwind acquired tax schemes and bring the tax structure in line with Sonovaʼs tax policies and BEPS (Base Erosion and Profit Shifting) principles.
- Full compliance: Sonova fully complies with the spirit and letter of local laws and regulations in alignment and within the frame of internationally recognized standards such as the OECD-G20 Inclusive Framework on Base Erosion and Profit Shifting (BEPS and BEPS 2.0) Initiatives as well as European guidelines (ATAD). If there are multiple options that provide equivalent business solutions, and that comply with all applicable laws, the most tax efficient approach is suggested, provided that the tax planning initiatives do not adversely impact the reputation of the Sonova Group.
- Transfer pricing: Sonovaʼs world-wide cross-border operations and added value chains are subject to yearly reviews to align Sonovaʼs Transfer Pricing Processes. Armʼs length profit allocation within the added value chains is granted through yearly reviews in line with multiple benchmarking analysis. Inter-company transactions are regularly monitored to ensure complete alignment with international standards and Sonovaʼs internal Transfer Pricing Processes.
- Cooperation with tax authorities: Although Sonova has not entered Advanced Pricing Agreements (APAs), it highly values open and proactive cooperation with tax administrations worldwide for any kind of tax matter. We continuously engage in constructive and transparent dialogue with tax authorities as part of our tax compliance policy.
Reporting
As mandated by the BEPS initiative, Sonova prepares a Country-by-Country Report (CbCR), filed with the Swiss Federal Tax Administration and thereby shared via automatic information exchange with tax authorities worldwide. The Transfer Pricing Master File is prepared by the Corporate Tax team, along with a master Local File. Local Files are completed by the local organizations (with support from the Sonova Corporate Tax team) according to OECD guideline Action 13 and Sonovaʼs Transfer Pricing Processes. Sonova is prepared to make its CbCR public once this becomes mandatory. As a multinational with a turnover above CHF/EUR 750 million, Sonova is also preparing for the requirements of a minimum tax under BEPS 2.0 – Pillar II.
Information on earnings before tax, reported taxes, reported tax rate, cash taxes paid, and cash tax rate is reported in the Note 5.1 Taxes in the 2022/23 Annual Report. Sonovaʼs tax rates might be lower than industry group averages because of largely uncapitalized group-wide net operating losses as well as net operating losses from previous periods in subsidiaries of acquired groups. Furthermore, Sonova is a Swiss-based multinational enterprise with significant activities, substance, risks, and assets in Switzerland, and the Swiss tax rate is lower than the global average tax rate. Cash tax paid is largely influenced by provisional advanced as well as final adjustment payments.
Sonova has also introduced a reporting tool to monitor, collect, and – where applicable – report information under Mandatory Disclosure Reporting, as introduced under EU Council directive 218/822 (DAC-6; enacted as of 2018). During the 2022/23 financial year, 110 cases were checked; in 12 of these Sonova has opted to report, in order to comply beyond any reasonable doubt with the formal requirements of the Directive.
Public policy
Donations to political parties
As a general rule, Sonova does not allow donations to political parties. This principle is stipulated in our Global Anti-Bribery Policy.
Association memberships
Sonova is an active participant in various associations and external initiatives to share specialist knowledge and to ensure high quality standards for hearing instruments and cochlear implants. We are a member of the following organizations, amongst others:
- Hearing Instrument Manufacturersʼ Software Association (HIMSA)
- European Hearing Instrument Manufacturers Association (EHIMA)
- Hearing Industries Association (HIA)
- Medical Device Manufacturers Association (MDMA)
- Hearing Instrument Manufacturersʼ Patent Partnership (HIMPP)
- Hearing Industry Research Consortium (IRC)
- European Telecommunications Standards Institute (ETSI)
We are represented in the governance bodies of certain organizations: CEO Arnd Kaldowski is the vice president of EHIMA; Stefan Launer, VP Audiology and Health Innovation, is Chair of HIMPPʼs Board of Directors; Victoria Carr-Brendel, President/GVP Cochlear Implants, is a board member of MDMA; and Sandra Brandmeier, President Sonova USA, is Vice Chair of HIAʼs Board of Directors. We are also represented in various professional and scientific associations related to core science and technologies relevant to our business.
In 2022/23, Sonova contributed around CHF 1.2 million in membership fees to trade associations and non-commercial organizations. Amongst the largest contributions (including membership fees) are those to the Hearing Industries Association (HIA), a forum for hearing aid manufacturers, suppliers, distributors, and hearing health professionals in the United States; European Hearing Instrument Manufacturers Association (EHIMA) and SwissHoldings, the federation of Swiss-based multinational enterprises.
Since 2016, Sonova has been a signatory to the UN Global Compact. This is a United Nations initiative that focuses on corporate citizenship, dialogue with stakeholders, partnerships, and communication. Sonova is part of both the global and local Swiss networks. Sonovaʼs CFO Birgit Conix is also a member of Accounting for Sustainability (A4S), a global network with the aim to inspire action by finance leaders to drive a fundamental shift towards resilient business models and a sustainable economy.
Animal welfare
As a manufacturer of medical devices. Sonova is required to demonstrate biological safety by complying with ISO 10993-1, the international standard for all medical devices with body contact. Under this standard, animal tests need to be considered in biological safety evaluations and, in some cases, cannot be completely avoided. In addition, as part of our contributions to research, Sonova provides components of cochlear implants to cochlear implant research centers and universities where they are tested on animals for basic research into, e.g., safety, feasibility, or efficacy of new technologies.
Sonova does not carry out any animal testing in-house and works with only a small number of third parties. We are committed to the “Three Rs” principle – replacement, reduction, and refinement – to limit animal testing as far as possible.
Replacement
- We use non-animal testing methods (in particular, testing with cell cultures) or chemical constituent testing where these methods are accepted by the respective regulatory bodies and can yield information as relevant as that obtained from in-vivo models.
- We promote the development and regulatory acceptance of new in-vitro methods by collaborating with test method developers and actively participating in working groups of the international standardization process.
Reduction
- We apply strategies to reduce the number of animals used in testing.
- We strive to avoid completely any unnecessary or duplicated testing by using previously evaluated or historically established biologically safe materials whenever possible and taking advantage of prior research among the various Sonova companies around the world.
- We emphasize risk assessment to clearly evaluate any need for animal testing.
Refinement
- We select whenever possible those test methods that minimize the distress caused to animals.
- We conduct all animal testing for biological safety evaluations through appropriately accredited testing laboratories in which all tests are carried out in accordance with good laboratory practice. We conduct animal testing for research collaborations only through universities and research laboratories where experiments are reviewed, approved, and overseen by the respective ethics committees.

